#ActionOnET
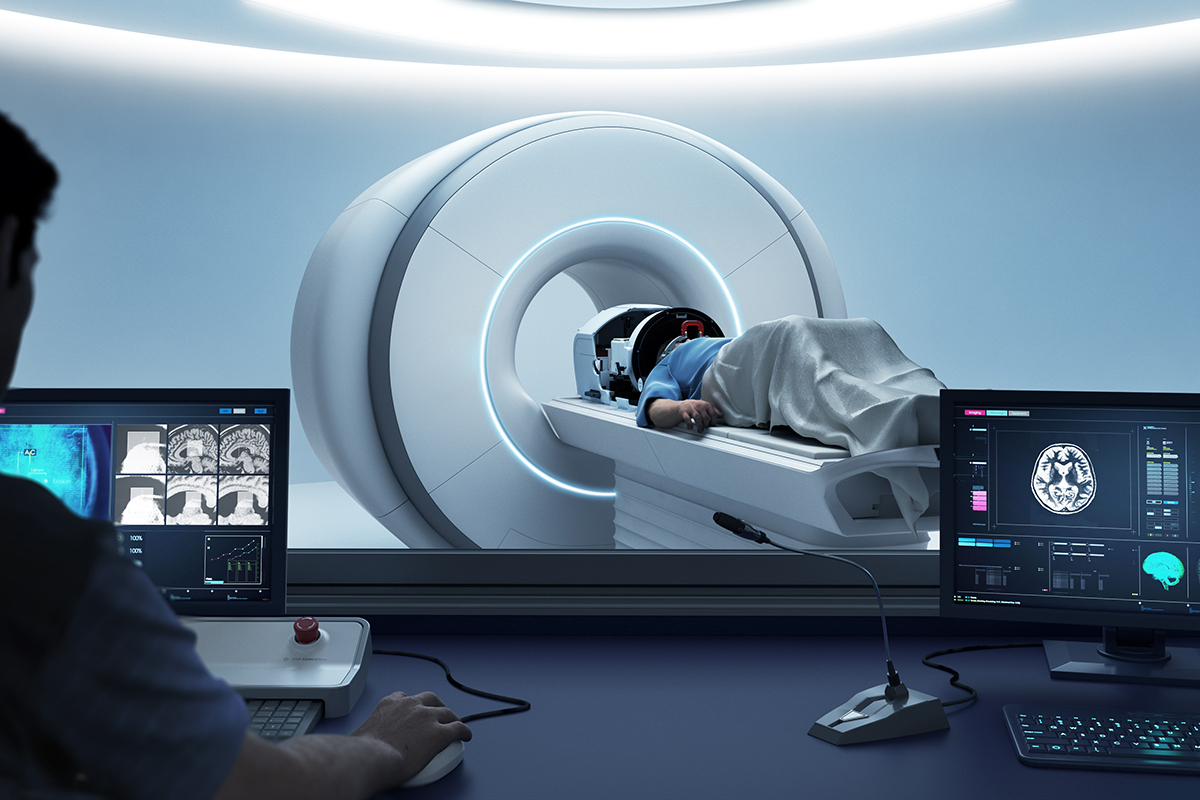
#ActionOnET is the leading UK campaign for Focused Ultrasound (MRgFUS) technology on the NHS for the treatment of essential tremor.
#ActionOnET is the leading UK campaign aimed at raising awareness and patient accessibility of Magnetic Resonance Guided Focused Ultrasound on the NHS for the treatment of Essential Tremor.
Essential tremor, which impacts over 1 million UK residents, can have a severe impact on quality of life, and patients often feel isolated as they are unable to do simple things like eat and drink in public.
However, Focused Ultrasound can be a life-changing treatment which NICE has ruled as being safe and effective. Making a significant improvement on people's lives.
The campaign aims to share the latest updates on MRgFUS with patients, the medical community, and policy makers, to transform lives of those living with the condition in the UK.
Follow the campaign
Follow us on Facebook, Twitter and Instagram and join in by using the #ActionOnET hashtag.
You can also sign up to our newsletter for the latest updates on #ActionOnET
Resources
Find out more
What can you do to help?
If having a NICE reviewed, innovative, effective, non-invasive treatment for ET available on the NHS is important to you, or people you know, then get in touch by using the contact form below, and we’ll let you know what you can do to make a difference.